Genome Wide Identification and Characterization of BRCA1 Genes inHomo Sapiens
Muhammad Waqar Mazhar1*, Hira Tahir2, Fatima Mazhar3, Saira Saif1, Javaria Mahmood1, Nibras1 and Mudasra Skindar1
1 Department of Biotechnology, Government College University Faisalabad, Faisalabad, Pakistan
2 Department of Bioinformatics and Biotechnology, University of Health Sciences, Lahore, Pakistan
3 Department of Microbiology, Muhammad Nawaz Sharif University of Agriculture, Multan, Punjab, Pakistan
- *Corresponding Author:
- Muhammad Waqar Mazhar
Department of Biotechnology,
Government College University Faisalabad,
Faisalabad,
Pakistan,
E-mail: waqarmazhar63@gmail.com
Received Date: April 19, 2021;Accepted Date: May 03, 2021; Published Date: May 10, 2021
Citation: Mazhar MW, Tahir H, Mazhar F, Saif S, Mahmoud J, et a l. (2021) Genome Wide Identification and Characterization of BRCA1 Genes in Homo Sapiens. J Mol Biol BiotechVol.6 No.3:01.
Abstract
Breast cancer is the most prevalent cancer in the world. Approximately one in every nine Pakistani women is likely to suffer from breast cancer showing an incidence rate of 50/100,000 and breast cancer is growing at an alarming rate in Pakistan. The main causative element remains unknown yet primary risk factors identified are sex, age, parity, genetics, lack of child bearing, breast feeding, higher hormonal levels individual lifestyle etc. In Indian and Asian women as compared with western women. Knowledge of this factor emphasizes the need to modify the timing of modalities of detection of early carcinoma and its management inherited mutations in the breast cancer susceptibility gene 1 (BRCA1) (MIM 113705) and breast cancer susceptibility gene 2 (BRCA2) (MIM 600185) are associated with a high risk of developing breast cancers in females of different age and ethnic group.
Keywords
Breast cancer; Carcinoma; Ubiquitination; Tumor; RNA Polymerase II; DNA Double Strand Breaks (DSBS)
Introduction
A BRCA mutation is a change in both BRCA1 and BRCA2, which are tumor silencer qualities. Many various kinds of changes in these qualities have been recognized, some of which have been resolved to be destructive, while others have no demonstrated effect. Unsafe changes in these qualities may deliver an inherited breast-ovarian malignant growth condition in influenced people. Just 5%-10% of bosom malignant growth cases in ladies are ascribed to BRCA1 and BRCA2 transformations with BRCA1 changes being somewhat more normal than BRCA2 changes [1]. The BRCA1 present on chromosome 17 q 21.13. The BRCA1 quality contains 22 exons, 110 kb of DNA. BRCA1 joins with other tumor silencers, DNA damage sensors and signal transducers to shape an enormous multi-subunit protein complex known as the BRCA1-Associated Genome Surveillance Complex (BASC). The BRCA1 protein partners with RNA polymerase II, and through the C-terminal area, likewise associates with histone deacetylase buildings. Subsequently, this protein assumes a part in record, and DNA repair of double strand DNA breaks ubiquitination, transcriptional regulation just as different functions.
BRCA1 and BRCA2 encode fundamental proteins for DNA Homologous Recombination Repair (HRR). Loss of capacity of either quality disables this high-fidelity DNA fix pathway and results in hereditary unsteadiness and an increased risk of breast cancer or ovarian malignant growth in germ line BRCA1/2 (gBRCA) transformation transporters. Imperfect HRR expands affectability of gBRCA-transformed tumors to DNA damaging agents including anthracyclines, platinum salts, or to novel agents that square equal DNA repair pathways, including poly (ADP-ribose) polymerase inhibitors (PARPi) [2]. PARP restraint impedes the maintenance of DNA single-strand breaks and results in slowing down of replication fork movement by catching PARP on the DNA break. Both add to the accumulation of DNA Double Strand Breaks (DSBs) that HRR-lacking cell can- not fix proficiently. Essential protection from PARPi in a subset of gBRCA patients restricts the capability of gBRCA status as the just biomarker of reaction to that of an improvement methodology. Also, acquired resistance in mono therapy responders is a challenge.
Past examinations utilizing in vitro models, transgenic mice and human tumor tests have outlined two kinds of obstruction components to PARPi in gBRCA cells: Free of HRR (cell expulsion of the PARPi, PARP1 loss, FANCD2 over articulation, SLFN11 inactivation or CHD4 loss) and Reliant on HRR recovery, either by BRCA-autonomous systems (deficiency of 53BP1, REV7/MAD2L2, PAXIP1/PTIP, Artemis) or by BRCA-dependent mechanisms. The later include secondary BRCA1/2 mutationsthat re-establish the reading frame and the expression of partially functional hypomorphic BRCA1 proteins (BRCA1-11q elective graft isoform, the RING-less BRCA1 generated by downstream interpretation inception, or HSP90-medi-ated adjustment of BRCA1 C-terminal freaks). Most working BRCA clinical samples has focused on ovarian cancer and has set up that HRR recovery through secondary BRCA1/2 mutations may act as an obstruction component to platinum salts and PARPi. Then again, little is thought about in vivo PARPi resistant mechanism in gBRCA BC [3].
Domains
The human BRCA1 protein comprises of four significant protein spaces; the Znf C3HC4-RING domain, two BRCT domains and the BRCA1 serine domain. These domains encode approx. 27% of BRCA1 protein. There are six known isoforms of BRCA1, with isoforms 1 and 2 containing 1863 amino acids each [4].
Zinc ring motif
The zinc RING motif, a Zn finger found in eukaryotic peptides, is 40-60 amino acids long and comprises of eight conserved metal-binding residues, two quartets of cysteine or histidine coordinate two zinc atoms. This motif contains a short antiparallel beta-sheet, two zinc-binding loops and a central alpha helix in a small domain. This RING domain collaborates with related proteins, including BARD1, which likewise contains a RING motif, to frame a heterodimer [5].
Serine cluster domain
BRCA1 Serine Cluster Domain (SCD) ranges amino acids 1280-1524. A portion of the domain is situated in exons 11-13. High rate of mutation occurs in exons. Revealed phosphorylation locales of BRCA1 are concentrated in the SCD, where they are phosphorylated by ATM/ATR kinases both in vitro and in vivo. ATM/ATR is kinases activated by DNA damage. Mutation of serine residues may influence localization of BRCA1 to sites of DNA damage and DNA damage response function.
BRCT domain
The dual repeat BRCT domain of the BRCA1 protein is an elongated structure approximately 70 Å long and 30-35 Å wide. The 85-95 amino acid domains in BRCT can be found as single modules or as multiple tandems repeats containing two domains. The dual tandem repeat BRCT domains are arranged in a head-to-tail-fashion in the three-dimensional structure, burying 1600 Å of hydrophobic, solvent-accessible surface area in the interface [6].
Materials and Methods
Sequence retrieval of BRCA1 gene
Retrieval of DNA and protein sequence of the gene BRCA1 in Homo sapiens from all NCBI databases, FASTA format of the BRCA1 protein and BLAST-P was run for Homo sapiens genome database to find similar sequences from protein databases. In order to perform this protein sequence (NP_009225.1) of Breast cancer gene 1 Homo sapiens (Humans) was given as query sequence. Target sequences then used in further databases like PFAM databases to find out the conserved domains and motifs.
Identification of domains and motifs
The sequence of BRCA1 protein was given as a query sequence and downloaded all target sequences in order to analyze the conserved motifs and domains of the protein sequences among (Homo sapiens), (Pan troglodytes) and (Gorilla gorilla) at NCBI online database Pfam and SMART. This step was done to remove sequences in which there were 4 conserved domains and motifs that are essential for BRCA1 protein to perform its functions normally. Only one conserved domain was identified in this Homo sapiens, smart00292, Cd00027, Cd00162, Pfam1282 that is a member of the superfamily. All of the variants were carefully examined with E-value less than 1 e- 10 and those with lengthy ORF were chosen for further data analysis using various databases, and Position of chromosome, length of protein and cDNA and genomic sequences were observed by using NCBI database.
Gene structure and phylogenetic analysis of TNF gene family
cDNA and genomic sequences were taken from NCBI and BRCA1 gene structure was observed by using an online tool named as Gene structure display sever to determine the position of introns and exons and three-dimensional structure of protein was obtained in BRCA1 gene. By using the bioinformatics tool MEGA7 with default parameters all the sequences from different organism like, (Homo sapiens), (Pan troglodytes) and (Gorilla gorilla) were aligned to perform multiplealignment and pairwise alignment in order to report the conserved domains in BRCA1 gene members. MEGA7 was used to coordinate sequences and a perfect unrooted evolutionary tree was built on the bases of alignments of sequences. Even after that, the BRCA1 gene family was classified based on their evolutionary similarity. A separate instrument was used to measure the molecular weight and isoelectric point toolbox. Evolutionary analysis was used to name the subfamilies of Breast cancer gene 1 based on their homologs. The evolutionary relationship among the families was performed using the online time tree server [7].
Results
There were 4 domains BRCA1 genes identified in Homo sapiens by using different tools and databases all the information was presented by a supplementary data table, such as RNA, DNA and proteins sequences. The position of gene was found on chromosome number 17. There are 22 exons and only 4 conserved domains were identified. Evolutionary relationship shows the similarity of different genes.
Homology analysis and phylogenetic analysis
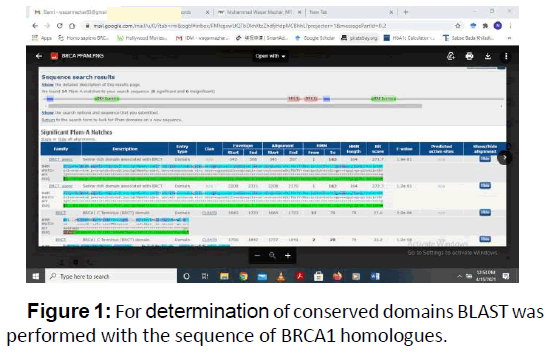
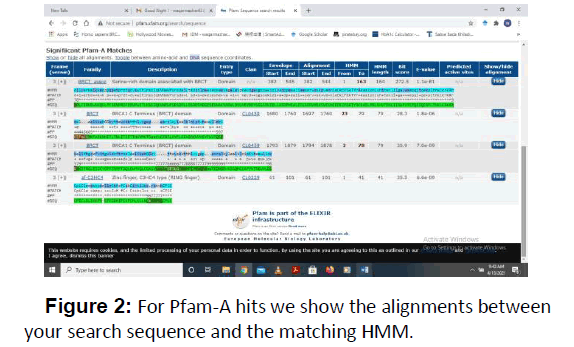
For determination of conserved domains BLAST was performed with the sequence of BRCA1 homologues. As a result of BLAST there were 4 conserved Smart 00292 Cd00027, Cd00162 and Pfam12820 which are the most conserved domain among BRCA1 protein members (Figures 1 and 2) (Table 1).
| Proposed names | BRCA1 |
|---|---|
| Gene Locus | MIM:113705 |
| Protein accession | 9225.1 |
| RNA accession | 7294.4 |
| Exons | 24 |
| Chr | 17 |
| ORF length | 5592 |
| Amino acid length | 1863 |
| Start of Genomic Location | NC_000006.12 |
| Conserved domains in protein sequence | Smart00292 Cd00027 Cd00162 Pfam12820 |
Table 1: S1 Proposed nomenclature and important features of BRCA1 genes
Homology and phylogenetic analysis
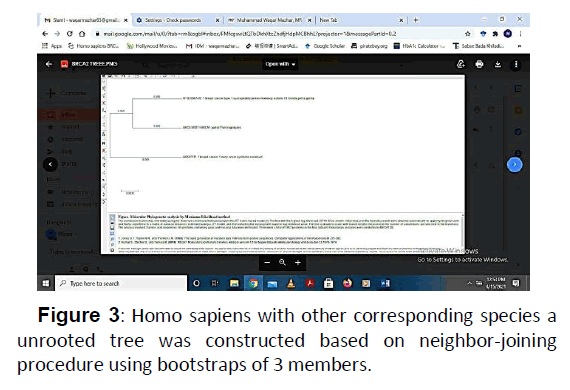
After that comparison of sequences of BRCA1 protein sequence was done with the identical sequences of different species (Homo sapiens), (Pan troglodytes), and (Gorilla gorilla) at NCBI. The result of this analysis revealed that there was high level of resemblance of BRCA1 protein with the other, but there was but pfam00229 was highly conserved in all the species. In order to determine the evolutionary relationship of BRCA1 Homo sapiens with other corresponding species a unrooted tree was constructed based on neighbour-joining procedure using bootstraps of 3 members (Figure 3).
Discussion and Conclusion
There are many different signalling molecules that are involved in mutation. There are also many signalling pathways and molecules involved in mutation regulation like growth factors especially on the downstream factor p53 and PTEN. BRCA1 was originally known as Tumour Suppressor, but the current studies have revealed that it is involved in diverse responses. Most important function performed by BRCA1 gene is to control the making a protein that act as tumour suppressor, it helps prevent cells from growing and dividing too rapidly or in an controlled way. Everyone has BRCA1 and BRCA2 gene. Some people have an inherited mutation in one or both of the genes that increase the risk of breast cancer; it can be passed to you from your parents.The exon-intron structure of the BRCA1 gene family was found to be very similar in this study. However, there was a wide range of gene distribution across chromosomes. The discovered genes could provide a wealth of knowledge for manipulating the BRCA1 genome in order to develop drugs.
References
- Chappuis P O, Nethercot V, Foulkes W D. (2000) Clinico–pathological characteristics of BRCA1‐and BRCA2‐related breast cancer. Semin. Surg. Oncol. 18:287–295.
- Beckerman M. (2005) Molecular and cellular signaling. Springer-Verlag New York.
- Cruz C, Bermejo M C, Enríquez S G, Guevara A L, Ibrahim Y H, et al. (2018) RAD51 foci as a functional biomarker of homologous recombination repair and PARP inhibitor resistance in germline BRCA-mutated breast cancer. Ann Oncol. 29:1203-1210.
- Gogola E, Rottenberg S, Jonkers J. (2019) Resistance to PARP inhibitors: lessons from preclinical models of BRCA-associated cancer. Annu Rev Cancer Biol. 3:235-254.
- Ibrahim, Majzoub F, Abdelaziz M S. (2015) Immunohistochemical Detection of EBV, BRCA1 and Ki67 in Triple Negative Breast Cancer. Sudan University of Science & Technology.
- Forties C E. (2008) Synthesis and Characterization of a Ru Coordinated AzobenzeneMetallofoldamer. The Ohio State University.
- Sinha S, Wang S M. (2020) Classification of VUS and unclassified variants in BRCA1 BRCT repeats by molecular dynamics simulation. ComputStructBiotechnol J. 18:723-736.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences